Medizintechnik
In der Medizintechnik vereint Codronic präzise Medizinrobotik und innovative Labortechnik mit fundierter Expertise in Regulatorik und Zulassungen. Wir entwickeln zukunftsweisende Lösungen für die bildgebende Diagnostik, die höchste Qualitätsstandards und normative Konformität im sensiblen Gesundheitssektor garantieren.
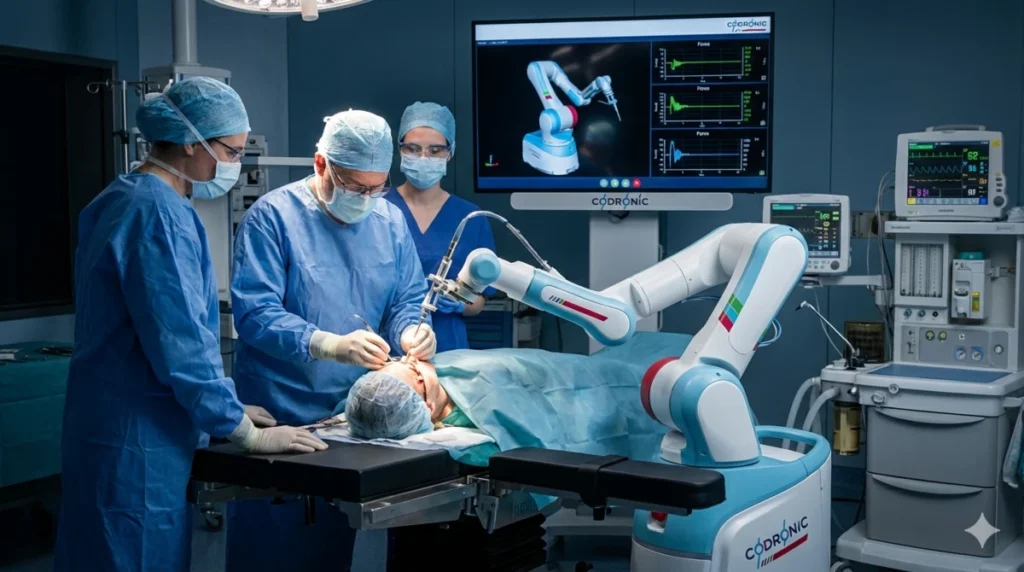
Medical Robotics
In medical robotics, we develop high-precision mechatronic systems that support surgeons during complex procedures. Our focus lies on the integration of sensitive sensors and actuators for haptic feedback as well as exact kinematics. Using state-of-the-art simulation methods such as FEM structural simulation, we ensure the necessary mechanical strength and absolute reliability of robotic arms in operation.
As an experienced development partner, we accompany the entire creation process according to the state of the art, from the initial concept study to the creation of high-quality manufacturing drawings. In doing so, we specifically consider the requirements for human-machine interaction in clinical environments. We provide all necessary technical verification procedures and seamless documentation for safety-critical functions and performance levels.

Diagnostic Imaging
Diagnostic imaging requires maximum mechanical precision and the interference-free integration of complex electronic components. We design innovative assemblies for MRI and CT systems as well as modern X-ray equipment under strict adherence to the tightest tolerances. Our deep expertise in special-purpose machinery enables the realization of high-precision positioning units that ensure the exact alignment of detectors and radiation sources in space.
Through topology optimization, we effectively reduce the weight of moving masses without compromising the required structural rigidity of the components. This significantly improves the dynamics of the diagnostic systems and increases the resulting image quality. Furthermore, we create standard-compliant technical documentation and perform detailed risk assessments to guarantee the safety of patients and users at all times.

Laboratory Technology
Maximum efficiency and the reproducibility of results are the core requirements of modern laboratory technology and automated analytical processes. We develop customized system solutions for liquid handling and sample preparation that are seamlessly integrated into existing customer processes. In doing so, we draw on extensive experience in cleanroom technology and the safe handling of sensitive assemblies.
Our mechatronic system development includes the targeted selection of suitable actuators for precise dosing tasks as well as the continuous optimization of process sequences. Through high-quality manufacturing drawings and structured technical documentation, we reliably secure the quality of the subsequent industrialization. We support our customers in designing innovative laboratory systems to be resource-efficient, sustainable, and absolutely future-proof.

Regulatory Affairs and Certification
Strict adherence to regulatory requirements forms the foundation for any successful market authorization in medical technology. We provide comprehensive support to medical device companies in creating technical documentation in accordance with the Medical Device Regulation (MDR) EU 2017/745, the In Vitro Diagnostic Regulation (IVDR) EU 2017/746, as well as relevant quality standards such as ISO 13485 and ISO 14971. Our services include detailed risk assessments and the professional evaluation of safety functions according to IEC 60601.
We perform auditable verification using FEM simulations to provide mathematically sound proof of the mechanical strength and safety of your medical devices. This methodical approach significantly accelerates the entire conformity assessment process. From the initial requirements list to the final operating manual, we ensure legally compliant documentation that also safely masters the transition to the new EU Machinery Regulation.



Your direct contact
Our team is here for you. We look forward to your inquiry.
FAQ
Wir erstellen die notwendige technische Dokumentation inklusive Risikobeurteilungen und Konformitätsbewertungen. Dabei begleiten wir den Übergang zur Maschinenverordnung (EU) 2023/1230 und sichern die Einhaltung aller normativen Anforderungen für eine erfolgreiche Zulassung.
Wir nutzen FEM-Struktursimulationen zur präzisen Festigkeitsanalyse und Topologieoptimierung Ihrer Komponenten. Diese prüffähigen Nachweise validieren die mechanische Sicherheit und optimieren den Materialeinsatz für hochpräzise Anwendungen in der Medizinrobotik oder Diagnostik.
Ja, wir entwickeln mechatronische Systeme und Sondermaschinen für die Automatisierung und Montage komplexer Baugruppen. Unsere Expertise umfasst die Integration von Sensorik sowie Aktorik und die Optimierung von Prozessen für sensible Reinraum-Umgebungen.
Durch die fundierte Bewertung von Sicherheitsfunktionen nach Performance Level und SIL garantieren wir höchste Zuverlässigkeit. Unsere Konstruktion erfolgt nach dem State of the Art, eingebettet in Ihre Kundenprozesse oder als eigenständiges Entwicklungsteam.
Unsere Spezialisten konzipieren kundenspezifische Maschinenlösungen gezielt für sensible Reinraum-Prozesse und anspruchsvolle Fertigungsumgebungen. Dabei kombinieren wir innovative Engineering-Methoden wie KI-gestützte Entwicklung mit Nachhaltigkeit und Ressourceneffizienz.
Hier finden Sie Blog-Beiträge zu unseren Engineering-Projekten

Fasteners – High-Tech in a Humble Guise

The Green Transformation is gaining ground

Mitigating the Skilled Labor Crisis with AI

Environmental Impact Assessment EIA

UAVs in 2030
